FEM-PrEP Closure: Is PrEP ready for the world?
(Updated April 19, 18:00) In July 2010 the world of HIV prevention felt the first tremor of hope that the fight against the virus that causes AIDS was at a turning point in its history after the pivotal CAPRISA trial (a “game changer”) showed that a topical gel containing an antiretroviral (ARV) used to treat HIV infection could reduce the risk of new infection by 39% in women.
In November 2010, a second wave shook the field after the landmark iPrEX trial showed that Truvada, a combination of two ARVs, could reduce the risk of HIV acquisition by 44% in men who have sex with men (MSM). With at least five more ongoing or planned trials, the hope was high that Pre-Exposure Prophylaxis (PrEP) could contribute to tame the HIV epidemic.
The CDC started producing guidance documents and the world preparing for PrEP implementation (or part of it) when the wave of warranted optimism on which the HIV Prevention field was surfing, suddenly and unexpectedly faltered on the 18th of April when the FHI-led trial FEM-PrEP announced its orderly closure: it’s Independent Data Monitoring Committee (IDMC) advised that the study was “highly unlikely to be able to demonstrate the effectiveness of Truvada in preventing HIV acquisition in the study population.” The IDMC advice was the results of a per protocol review of safety and effectiveness of the product as well as a further analysis of the data. Based on the feedback of the IDMC, the investigators decided to proceed with orderly closure of the trial due to futility.
The wording here is important to both clarify and understand: the premature closure of the FEM-PrEP trial does not signal the end of hope or of other PrEP studies. The FEM-PrEP study results do not show that Truvada does not work for preventing HIV acquisition, only that if this study would have continued as planned, “it would be highly unlikely that [it] would have been able to conclude that Truvada was effective to prevent HIV infection in this population of women” as explained by FHI Tim Mastro. “There was no good reason to continue” he added during an advocate call held by AVAC.
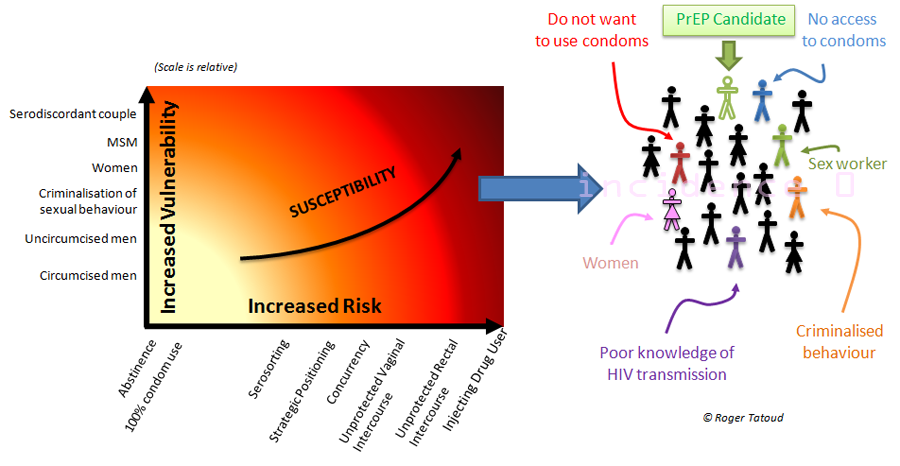
There are many reasons to explain that result; that the drug did not work for that particular purpose is one of them, but that participants were not taking the drug as prescribed, that there may be biological differences between men and women leading to different efficacy, that the route of exposure matters or that there was a difference in the composition of the study groups are some of the others possible reasons.
It could also be that, for reasons such as risk reduction conselling and provision of condoms or because the point estimate of efficacy was set too high (i.e. how well Truvada was expected to work for preventing new infections), the trial no longer had the power to detect a statistical difference between the two groups. In this case the trial can not answer the question it was asking so that “at this time, it cannot be determined whether or not Truvada works to prevent HIV infection in women.” (FHI Statement).
Very little is yet known about the study results and trying to figure out answers without data is nothing but futile speculations and unwarranted assumptions. But that there was more pregnancies in the group of women receiving the ARV drug than in the group given the placebo (an inactive compound) is one remarkable observation. There is no data to suggest that Truvada impacts on pregnancy rate so far but this observation definitively requires further analysis.
Pregnancy rates are telling us more. Women enrolled in the study were required to use an effective method of contraception and should not want to conceive during the time they would stay in the study. At enrolment, when the study started, 66% of the participants were using injectables and 30% oral contraceptives. Women not using any contraception were counselled to receive one of the study-approved methods that was then provided for free.
However, despite 100% of the women allegedly using a form of contraception, 9% became pregnant with, unsurprisingly, a higher pregnancy rate in women using oral contraception. This tells us two important things at this stage of the research on the use of PrEP for HIV prevention:
- That despite all good intentions, taking a pill a day is not necessarily easy with the corollary that not taking it as prescribed has consequences,
- That a minimum level of adherence is required and need to be maintained for an intervention to be successful.
But we are not talking about Truvada pills here; we are talking about contraceptive pills (and injectables). This observation about a “parallel intervention” should inform the future PrEP developments, which so far requires oral daily dosing in MSM and twice daily dosing in women (before and after sex). If taking one pill a day is already a challenge (contraceptive), what about taking two pills a day (contraceptive and ARV)?
Adherence has long been recognised as a major and crucial factor in the success of PrEP (as it is with many medical interventions). Adherence level in iPrEX study was at best average with drug detection indicating less than daily dosing in 40% to 50% of the participants. On the other hand, near perfect adherence was reported in the Partners-PrEP study which enrolled sero-discordant couples. Adherence in the FEM-PrEP study has been recorded using a number of approaches including self-reported adherence, pill counts and blood test. Participants also received a participant-centred and goal-orientated counselling and enhanced adherence counselling when needed. Much was done to ensure that adherence would not fail the trial and we now have to wait for the full data to confirm the preliminary reported 95% adherence (based on self-reporting. Final adherence figure is expecetd to be lower).
Conversely, the number of new HIV infections (56 in total, equaly distributed between the Truvada and placebo groups) emphasises that despite regular counselling and provision of condom, unprotected sex was happening, contributing to a 5% estimated incidence similar to that observed in other similar trials. Providing free condoms probably contributed to a decrease in the number of new HIV infections (assumption to be confirmed) and potentilaly loss of trial power, but the study could not demonstrate that adding Truvada could improve the outcome of the trial for reasons yet to be determined. However, this underlines that Combination Prevention, which is now the motto at the heart of HIV prevention programming, may be more complicated than just stacking up or alternating existing or forthcoming interventions.
Where do we stand now? As AVAC’s Mitchell Warren said, this result “must be seen as what it is – the closure of a single trial in a field that has generated exciting results in the recent past. Even with this finding, there is still a strong rationale for continuing other trials, including those in women, in hopes of obtaining better results in the future.”
The debate about the future of Truvada for HIV prevention continues with those who oppose to it feeling vindicated by the closure of FEM-PrEP. For other, it is the confirmation that “science does not always produce the answers we hope for”. However, curbing slightly the enthusiasm that followed the landmark iPrEX and pivotal CAPRISA – an enthusiasm that echoes a real need for more HIV prevention strategies, is an opportunity to take stock, to reflect and engage with all stakeholders and even listen to dissenting voices, and to keep in mind that if haste is generally the enemy of perfection, when it comes to medical research, haste is the enemy of quality.
We now must wait for the full results to be available and then, in conjunction with the results from past and ongoing PrEP studies, strategise the next move, onwards.
rjt
| Notes:For more information about the study, see the following fact sheets available on the FHI website About FEM-PrEP, Key Findings , Questions and Answers, AdherencePregnancy and Contraceptive Effectiveness in the FEM-PrEP Trial
NAM aidsmap has published an informative summary of the available results online. This post was updated to reflect new information communicated during an AVAC call on Tuesday 19 April 2011. |




Great article, thank you. One of the shocking things about the trial, though, is that incidence, the annual rate of new infections, is extremely high. 5% incidence, said to be similar to the figure for other trials, would result in a very serious epidemic, like those in Swaziland, Lesotho or South Africa, not like those in Kenya, Tanzania or Uganda.
Of course, the trial population was chosen because incidence is very high, but the fact that this large group of people were still at very high risk of HIV infection, despite being counselled, provided with condoms and regularly being followed up, suggests that there may be more to HIV transmission than sexual behavior. That could mean that pills, even those with high effectiveness and in very large quantities, may not have much impact on HIV transmission.
Trials like this show that we don’t know, or don’t accept, exactly how HIV is being transmitted in very high prevalence areas. PrEP, microbicides and even condoms all presuppose sexual transmission. The failure of rigorous counselling (or the most rigorous that we currently know of) and other precautions may suggest that we should investigate other modes of HIV transmission.
PrEP may eventually be able to reduce sexual transmission of HIV, but it will still have no impact on non-sexual transmission. To improve trial results, trials could start to ensure that transmissions that occur really are sexual transmissions and that non-sexual transmissions are excluded from trial results. ‘Efficacy’ would instantly be improved if they could do that for any of the previous trials, including the FEM-PrEP trial.